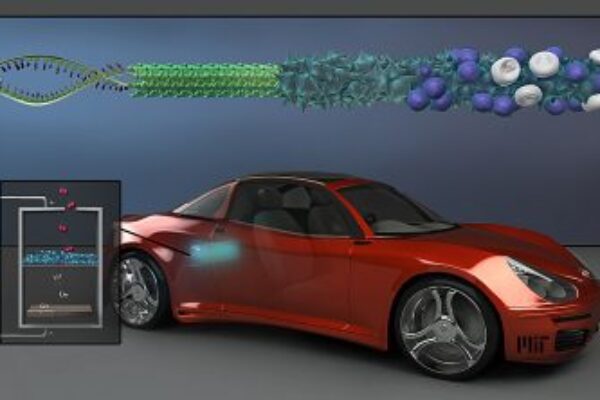
MIT researchers use modified viruses to boost lithium-air battery performance
The key to research study was finding a way to modify the nanowires – wires that are about the width of a red blood cell, and can serve as one of a battery’s electrodes – to increase the area where electrochemical activity takes place during charging or discharging of the battery.
The researchers produced an array of nanowires, each about 80 nm across, using a genetically modified virus called M13, which can capture molecules of metals from water and bind them into structural shapes. In this case, wires of manganese oxide were used to create a lithium-air battery’s cathode were made by the viruses. Unlike wires ‘grown’ via conventional chemical methods, the virus-built nanowires have a rough, spiky surface that increases their surface area.
Angela Belcher, the W.M. Keck Professor of Energy and a member of MIT’s Koch Institute for Integrative Cancer Research, explained that the biosynthesis process is “really similar to how an abalone grows its shell”.
The increase in surface area produced by this method can boost the lithium-air batteries’ rate of charging and discharging. But the process also has other potential advantages. "Unlike conventional fabrication methods, which involve energy-intensive high temperatures and hazardous chemicals, this process can be carried out at room temperature using a water-based process," explained Belcher.
Also, rather than isolated wires, the viruses naturally produce a three-dimensional structure of cross-linked wires, which provides stability for an electrode.
A final part of the process is the addition of a small amount of a metal, such as palladium, which greatly increases the electrical conductivity of the nanowires and allows them to catalyze reactions that take place during charging and discharging. Other groups have tried to produce such batteries using pure or highly concentrated metals as the electrodes, but this new process drastically lowers how much of the expensive material is needed.
The modifications have the potential to produce a battery that could provide two to three times greater energy density – the amount of energy that can be stored for a given weight – than today’s best lithium-ion batteries.
Belcher emphasizes that this is early-stage research, and much more work is needed to produce a lithium-air battery that’s viable for commercial production. This work only looked at the production of one component, the cathode; other essential parts, including the electrolyte – the ion conductor that lithium ions traverse from one of the battery’s electrodes to the other – require further research to find reliable, durable materials. Also, while this material was successfully tested through 50 cycles of charging and discharging, for practical use a battery must be capable of withstanding thousands of these cycles.
While these experiments used viruses for the molecular assembly, Belcher says that once the best materials for such batteries are found and tested, actual manufacturing might be done in a different way. This has happened with past materials developed in her lab, she says: The chemistry was initially developed using biological methods, but then alternative means that were more easily scalable for industrial-scale production were substituted in the actual manufacturing.
In addition to Oh, Belcher, and Shao-Horn, the work was carried out by MIT research scientists Jifa Qi and Yong Zhang and postdoc Yi-Chun Lu. The work was supported by the U.S. Army Research Office and the National Science Foundation.
The new research is described in a paper published in the journal Nature Communications, co-authored by graduate student Dahyun Oh, professors Angela Belcher and Yang Shao-Horn, and three others.
More information about the research work at
https://web.mit.edu/newsoffice/2013/better-batteries-through-biology-1113.html
 If you enjoyed this article, you will like the following ones: don't miss them by subscribing to :
eeNews on Google News
If you enjoyed this article, you will like the following ones: don't miss them by subscribing to :
eeNews on Google News



